|
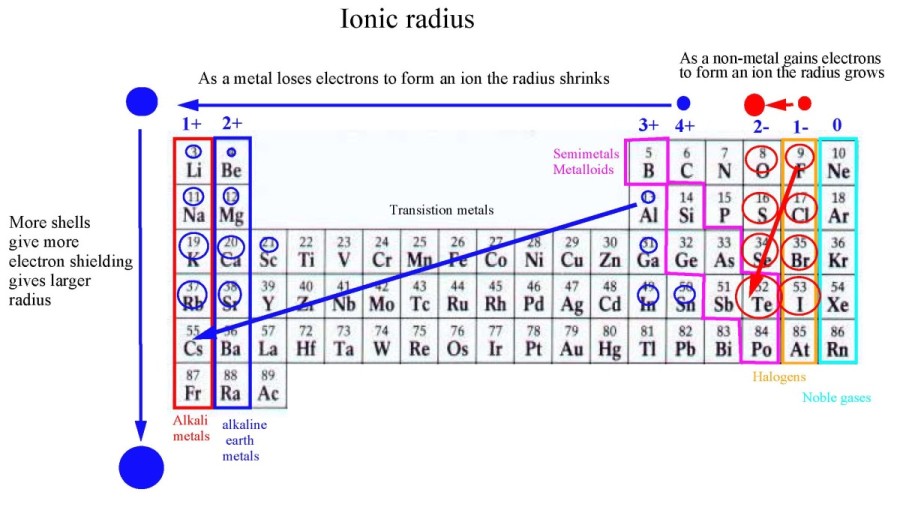
An atom having such an electronic structure tends to form a stable shell of eight electrons by adding two more, producing an ion that has a double negative charge. The elements belonging to Group 16 of the periodic table are characterized by electron configurations in which six electrons occupy the outermost shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
